You’ve likely heard about medical & Pharma breakthroughs during wartime, but the story of how a deadly chemical weapon became the foundation of modern cancer treatment remains one of medicine’s most remarkable transformations. This guide is for healthcare professionals, medical students, history enthusiasts, and anyone curious about how wartime pharmaceutical research led to life-saving discoveries.
Your journey through this medical breakthrough will reveal three incredible developments. First, you’ll discover how a tragic 1943 bombing in Bari, Italy accidentally exposed the cancer-fighting potential hidden within mustard gas. Then, you’ll learn about the first human chemotherapy trial that made history when Yale researchers took an experimental leap with a terminal patient known only as “JD.” Finally, you’ll explore how these wartime medical discoveries transformed into the sophisticated cancer treatments saving lives today.
From chemical weapons to cancer treatment breakthrough – this transformation shows how even the darkest chapters of war can lead to humanity’s greatest medical victories.
The Tragic Discovery That Changed Medicine Forever
The Bari Harbor Bombing and Its Deadly Aftermath
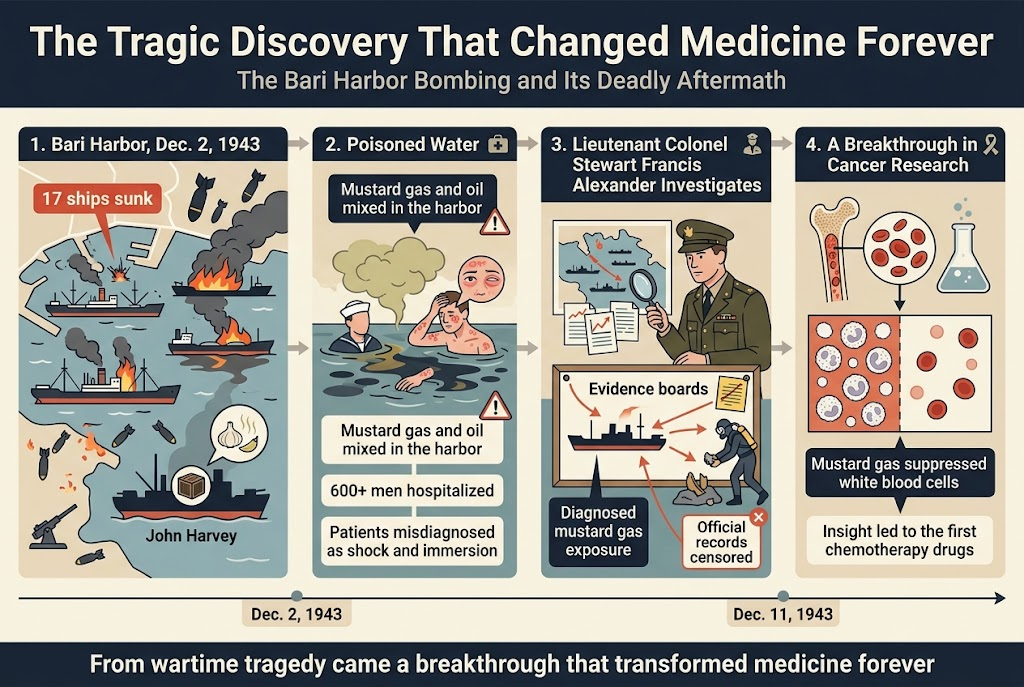
On December 2, 1943, you would have witnessed one of the most devastating yet transformative moments in wartime medical discoveries at the harbor of Bari, Italy. The Allied forces had packed this strategic port with oil tankers and cargo ships, believing themselves safe from German attack. Your commanders considered the Luftwaffe severely depleted, deploying only a single anti-aircraft battery to defend the entire city.
However, recognizing this enormous opportunity, the German air force struck at dusk with over 100 warplanes. You would have seen German bombs ignite catastrophic explosions among the closely anchored vessels laden with fuel and ammunition. Seventeen ships were sunk in what became known as the “second Pearl Harbor.” But as you’ll discover, the true horror was just beginning.
During the attack, hundreds of men struggling in the contaminated water noticed something alarming—a distinctive pungent odor resembling garlic or mustard. You might wonder what could cause such a smell during a bombing raid. The answer was classified at the highest levels: one American Liberty ship, the John Harvey, carried a secret cargo of 2,000 mustard gas bombs.
When German bombs struck this vessel, you would have witnessed a poisonous cloud wafting over the city while toxic fluid coated hundreds of sailors swimming desperately for their lives. The chemical spread insidiously throughout the harbor, mixing with tons of fuel oil from the damaged ships, creating a deadly cocktail that would change pharmaceutical research forever.
Over 600 men were hospitalized with what you would initially observe as mysterious symptoms. In the crushing chaos of that first night, medical personnel mistakenly believed survivors were suffering only from shock and immersion. You would have seen patients given morphine, wrapped in warm blankets, and left sitting in their oil-soaked uniforms for 12 to 24 hours while more obviously wounded casualties received priority treatment. This unknowing exposure was tantamount to marinating in mustard gas.
By dawn, you would have witnessed patients developing red, inflamed skin and blisters “the size of balloons.” Within 24 hours, hospital wards filled with men whose eyes had swollen completely shut. The medical staff suspected some form of chemical irritant, but you would notice these patients didn’t present typical symptoms or respond to standard treatments. The situation grew more ominous when headquarters classified these burn patients with unusual symptoms as “Dermatitis N.Y.D.”—not yet diagnosed.
Lieutenant Colonel Alexander’s Investigation and Crucial Findings
Without warning, you would have observed patients in relatively good condition beginning to die mysteriously. These sudden deaths left doctors baffled and desperate for answers. Rumors spread that Germans had deployed an unknown poison gas, and with daily death tolls rising, British officials placed a “red light” emergency call to Allied Force Headquarters in Algiers.
Your military commanders dispatched Lieutenant Colonel Stewart Francis Alexander, a young chemical warfare specialist attached to Eisenhower’s staff, immediately to investigate this medical crisis. Despite British port authorities’ denials, you would have witnessed Alexander quickly diagnosing mustard gas exposure through his methodical investigation.
Alexander pursued his investigation with remarkable determination, convinced that military security preoccupation had compounded the tragedy. You can trace his scientific approach as he carefully studied medical charts and plotted the destroyed cargo ships’ positions relative to gas victims. His meticulous work succeeded in pinpointing the John Harvey as the epicenter of the chemical explosion.
When divers retrieved fragments from the harbor floor, you would learn these fractured shell casings were identified as 100-pound American mustard bombs. On December 11, 1943, Alexander informed headquarters of his shocking findings: not only was the gas from the Allies’ own supply, but victims labeled “Dermatitis N.Y.D.” had suffered prolonged exposure from being immersed in the toxic mustard and oil solution floating on the harbor’s surface.
The response Alexander received would astound you. While General Eisenhower accepted his diagnosis, Prime Minister Churchill refused to acknowledge mustard gas presence in Bari. Your Allied leaders, with the war entering a critical European phase, imposed strict censorship on this chemical disaster. All mention of mustard gas was stricken from official records, and Alexander’s diagnosis was deleted from medical charts.
How Mustard Gas Exposure Revealed Unexpected Medical Properties
Now that you understand the cover-up’s scope, you’ll discover how Alexander made another surprising observation that would revolutionize cancer treatment. He noticed his patients’ white blood cell counts were extremely low, leading him to deduce that mustard gas had damaged patients’ bone marrow—the very source where white blood cells are produced.
This wartime medical discovery sparked a remarkable insight. Alexander began thinking: if mustard gas could suppress white blood cell production so dramatically, what potential could it hold for treating leukemia? You should understand that leukemia represents a form of cancer characterized by dangerous white blood cell overproduction—essentially the opposite problem these victims faced.
Your understanding of this pharmaceutical breakthrough deepens when you realize Alexander’s observation connected a chemical weapon’s devastating effects to potential therapeutic applications. The toxic agent that had caused so much suffering in Bari harbor revealed unexpected medical properties that could potentially combat cancer’s uncontrolled cell growth.
This tragic discovery would soon transform from a wartime disaster into one of the most significant advances in chemotherapy development timeline. Alexander’s investigation, despite being censored and hidden from public view, laid crucial groundwork for future cancer treatment breakthroughs. His scientific observations about mustard gas effects on bone marrow and white blood cell production became the foundation for developing the first generation of cancer-fighting drugs.
The Bari harbor incident demonstrates how wartime medical discoveries often emerge from the most unlikely and tragic circumstances, forever changing our approach to treating life-threatening diseases.
From Chemical Weapon to Cancer Treatment Breakthrough
Alexander’s Groundbreaking Animal Studies on Nitrogen Mustard
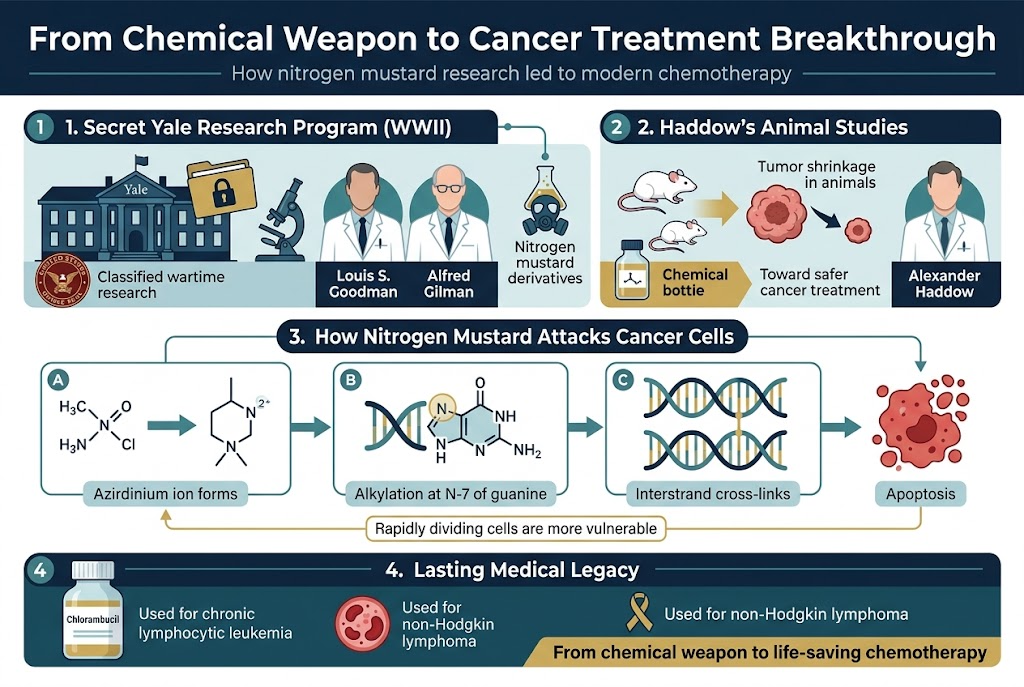
When you explore the transformation of chemical weapons into cancer treatments, you’ll discover that Alexander Haddow’s pioneering animal studies laid the crucial groundwork for modern chemotherapy. After World War II ended, you can see how British chemist Alexander Haddow continued the research that had been initiated during wartime, focusing specifically on identifying which chemical structures in nitrogen mustard could be modified to make the compound safer and more effective for medical use.
Haddow’s meticulous work with laboratory animals revealed something remarkable – you could observe how nitrogen mustards demonstrated a unique ability to target rapidly dividing cells. His studies showed that when administered to animals with tumors, these compounds caused significant tumor shrinkage, suggesting their potential to specifically target cancerous cells while minimizing damage to healthy tissue.
Through his systematic approach, you can understand how Haddow identified the precise molecular mechanisms that made nitrogen mustards effective against cancer. His animal studies demonstrated that these compounds worked by forming cyclic ammonium ions that then alkylated DNA, specifically attacking the N-7 nucleophilic center on guanine bases. This process created interstrand cross-links that forced cancer cells into apoptosis, effectively destroying them.
Most significantly, Haddow’s research led to the development of chlorambucil, a breakthrough treatment that you can still find in use today for chronic lymphocytic leukemia and non-Hodgkin lymphoma. His work essentially marked the birth of modern oncology, transforming wartime chemical research into life-saving medical applications.
The Secret Yale University Research Program During WWII
During the height of World War II, you would have found one of the most secretive and groundbreaking medical research programs operating within the walls of Yale University. Under the leadership of pharmacologist Louis S. Goodman and physiologist Alfred Gilman, this classified program focused on studying nitrogen mustard derivatives as potential defensive measures against chemical warfare.
What you need to understand about this program is that it operated under strict military secrecy. The Yale researchers were tasked with understanding how to defend against chemical weapons, but their animal studies revealed something unexpected and revolutionary. When they administered nitrogen mustard to laboratory animals, they observed that tumors shrank dramatically, suggesting that these compounds could target rapidly dividing cancer cells.
In December 1942, you can trace the moment when this secret research transitioned from defensive warfare studies to offensive cancer treatment. The Yale team initiated classified human clinical trials of nitrogen mustards for lymphoma treatment – marking the beginning of what you now know as chemotherapy. This represented the first systematic attempt to use chemical compounds specifically designed to fight cancer.
The secrecy surrounding this program was so complete that you wouldn’t have found any public documentation of their work until after the war ended. The researchers operated under military contracts, conducting studies that would fundamentally change how you understand cancer treatment today. Their groundbreaking work remained classified because of its potential military applications, but the medical implications were becoming increasingly clear.
Understanding How Chemical Agents Attack White Blood Cells
Now that you understand the research background, you need to grasp the precise biological mechanisms that made nitrogen mustards effective against cancer. When you examine how these chemical agents work, you’ll discover they operate through a sophisticated process of DNA alkylation that specifically targets white blood cells.
You can observe that nitrogen mustards form cyclic ammonium ions (aziridinium ions) through intramolecular displacement of chloride leaving groups by amine nitrogen atoms. Once these aziridinium groups form, they alkylate DNA by attacking the N-7 nucleophilic center on guanine bases. What makes this process particularly effective is that a second attack occurs after displacement of the second chlorine atom, forming interstrand cross-links (ICLs) between DNA strands.
During wartime observations, you could see that mustard gas destroyed bone marrow and lymphatic tissue – the very organs essential for producing immune cells. Researchers like Goodman and Gilman reasoned that this destructive property could be redirected to suppress cancerous white blood cells, laying the foundation for chemotherapy as you know it today.
The cytotoxic effect you observe comes specifically from the formation of these interstrand cross-links. These lesions force cells to undergo apoptosis via p53, a protein that scans the genome for defects. What’s crucial for you to understand is that the alkylating damage itself isn’t directly cytotoxic – rather, it’s the cellular response to this damage that leads to cancer cell death.
You’ll find that nitrogen mustards are nonspecific DNA alkylating agents, meaning they don’t distinguish between healthy and cancerous cells initially. However, because cancer cells divide more rapidly than most healthy cells, they’re more susceptible to this type of DNA damage, making them primary targets for destruction while allowing healthy tissue to recover more effectively.
The First Human Chemotherapy Trial That Made History
Patient JD’s Desperate Situation and Willingness to Try Experimental Treatment
When you examine the remarkable story of patient J.D., you’re looking at a man whose desperation became the catalyst for chemotherapy development timeline that would change cancer treatment forever. By August 1942, J.D.’s condition had deteriorated to an almost unimaginable state. You can picture the 47-year-old Polish immigrant, living alone in his rented four-room house in Meriden, Connecticut, facing a tumor mass so massive and hard that he couldn’t even turn his head.
J.D.’s journey into cancer treatment history began in February 1941 when what started as enlarged tonsils had evolved into full-blown lymphosarcoma – a malignant tumor of his lymph nodes. Throughout 1941, you would have witnessed doctors attempting to shrink his tumors with radiation therapy, but these conventional treatments provided no significant benefit. The cancer continued its relentless advance.
When J.D. was admitted to New Haven Hospital on August 25, 1942, his physical state was truly dire. You need to understand that his tumor had grown so large that it severely compromised his basic life functions. He couldn’t eat properly, couldn’t sleep comfortably, couldn’t breathe well, and was essentially trapped in his own body by the cancer that was choking off his ability to live normally.
What makes J.D.’s story particularly poignant is his isolation. He had immigrated to the United States at age 18, never married, had no known family around him, and worked in a ball-bearing factory until his illness forced him to stop in 1940. When Dr. Gustaf Lindskog approached him about participating in an experimental treatment with an unnamed chemical – what we now know was nitrogen mustard – J.D. faced this momentous decision entirely alone.
Despite knowing he was dying and understanding that this experimental treatment came with unknown risks, J.D. willingly agreed to become part of medical history. His courage in agreeing to this first chemotherapy trial stemmed from his recognition that conventional treatments had failed him completely, and this experimental approach represented his only remaining hope.
The Yale Team’s Bold Decision to Test Nitrogen Mustard on Humans
Now that we’ve covered J.D.’s desperate circumstances, you need to understand the extraordinary scientific leap that the Yale team was taking. The decision to move from animal testing to human trials represented one of the most significant moments in wartime medical discoveries.
During the early years of World War II, you should know that Yale School of Medicine researchers were participating in a top-secret U.S. government program. Their original mission was developing antidotes to chemical weapons to cancer treatment agents, as the government feared Germans would deploy these weapons against Allied troops. However, the Yale team’s attention had turned to nitrogen mustard for an entirely different purpose.
The preclinical work that led to J.D.’s treatment began with a collaborative effort between Drs. Louis Goodman and Alfred Gilman from the pharmacology department and Dr. Thomas Dougherty from anatomy. When they treated a mouse with advanced lymphosarcoma using nitrogen mustard, you would have witnessed something remarkable – after just two injections, the tumor began to soften and reduce in size.
What made their decision particularly bold was that they had observed both the promise and the limitations of nitrogen mustard in animal studies. While the initial results were encouraging, they also discovered that after another round of treatment, the tumor began growing again and the nitrogen mustard proved ineffective – the cancer had developed resistance. Despite this limitation, the fact that chemotherapy had extended the mouse’s life for 84 days was considered remarkable for that era.
The Yale physicians felt their data were encouraging enough to justify a human trial, leading them to approach Dr. Gustaf Lindskog, an assistant professor of surgery. The team needed to find a suitable cancer patient for this first chemotherapy trial, and J.D. became their candidate.
Due to wartime secrecy and censorship requirements, you need to understand that the Yale team couldn’t even document what they were doing openly. In J.D.’s medical records, they couldn’t note that he was receiving nitrogen mustard – instead, they referred to the treatment as “a lymphocidal chemical” or “substance X.” This secrecy added another layer of complexity to an already unprecedented medical undertaking.
Remarkable Initial Results and Eventual Treatment Resistance
With this experimental treatment underway, you can now witness how J.D.’s case demonstrated both the incredible promise and sobering limitations that would define chemotherapy development timeline for decades to come.
The initial results were nothing short of extraordinary. By August 31, just four days after receiving his first injection of nitrogen mustard, J.D. was showing clear signs of improvement. You would have observed him sleeping comfortably in bed for the first time in weeks, eating more easily, and regaining the ability to move his head in a wider arc. Most remarkably, he could cross his arms on his chest – a simple gesture that had been impossible due to his massive tumor burden.
By September 6, J.D.’s condition had improved so markedly that it must have seemed miraculous to the Yale team. However, you need to understand that this dramatic improvement came with concerning side effects. His white blood count, which indicates the levels of disease-fighting white blood cells, was falling precipitously – an early indication of the bone marrow suppression that would become a hallmark challenge of chemotherapy.
The most remarkable milestone came a month into treatment: J.D.’s cancer became undetectable. This represented the first documented complete response to intravenous chemotherapy in human cancer treatment history. You’re witnessing the moment when the concept of using chemicals to fight cancer moved from theoretical possibility to proven reality.
Unfortunately, whatever cancer cells remained had developed resistance to the nitrogen mustard, just as the Yale team had observed in their mouse studies. J.D.’s lymphosarcoma returned with devastating force. You would have witnessed his rapid deterioration as he suffered from bone marrow depression, bleeding, and respiratory distress. On December 1, 1942, after 96 days in the hospital, J.D. died.
Despite the tragic outcome, J.D.’s case established fundamental principles that continue to guide cancer treatment today. His experience revealed both chemotherapy’s effectiveness against cancer and its potentially lethal side effects, including bone marrow depression. As Dr. John Fenn noted when the medical records were finally recovered nearly 70 years later, “It was all there. In one patient, there was the revelation that cancer would respond to chemical injections, and that chemotherapy also had potentially lethal implications.”
This first chemotherapy trial became the foundation upon which modern oncology was built, proving that cancer could respond to chemical treatment while simultaneously revealing the ongoing challenges of toxicity and resistance that researchers continue to address today.
How Wartime Research Transformed Modern Cancer Care
The Development and FDA Approval of Mechlorethamine
With this in mind, next, we’ll see how the wartime discovery of nitrogen mustard transformed into a legitimate pharmaceutical breakthrough. You’ll find that mechlorethamine, commonly known as nitrogen mustard, underwent a remarkable journey from chemical weapon to FDA-approved cancer treatment.
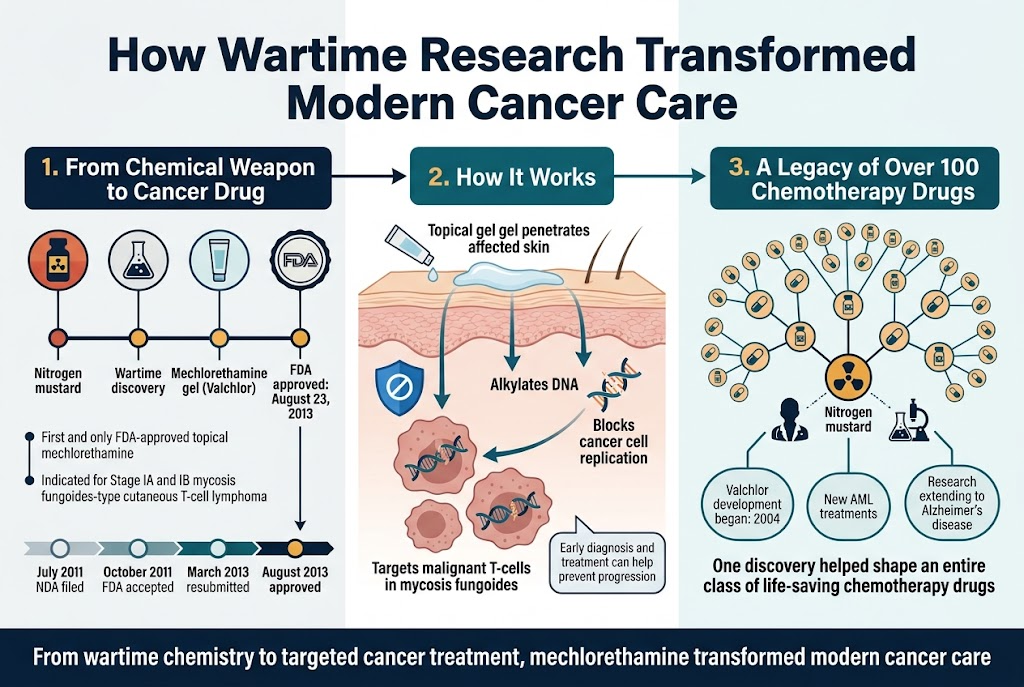
The development timeline for mechlorethamine as a modern therapeutic agent demonstrates how wartime medical discoveries evolved into sophisticated treatments. You can trace this evolution through the work of researchers like Professor Peter Crooks, Ph.D., who helped develop Valchlor, a breakthrough gel formulation of mechlorethamine. Beginning his developmental work in 2004, Crooks faced significant challenges in creating a stable topical formulation that could deliver the drug effectively while preventing systemic absorption.
When you examine the FDA approval process, you’ll discover that Valchlor (mechlorethamine gel) received its first approval on August 23, 2013, marking a historic milestone. This alkylating agent became the first and only FDA-approved topical formulation of mechlorethamine, specifically indicated for the topical treatment of Stage IA and IB mycosis fungoides-type cutaneous T-cell lymphoma.
The development journey you’re witnessing here spans nearly a decade of dedicated research. Ceptaris Therapeutics, Inc., the company behind Valchlor, navigated through multiple regulatory submissions, including the initial New Drug Application (NDA) filing in July 2011, FDA acceptance in October 2011, a resubmission in March 2013, and final approval in August 2013.
Understanding DNA Alkylation and Cancer Cell Destruction Mechanisms
Now that we have covered the regulatory pathway, you need to understand how mechlorethamine actually destroys cancer cells. As an alkylating agent, mechlorethamine works by targeting the DNA of malignant cells, particularly those problematic T-cells that characterize cutaneous T-cell lymphoma.
When you apply Valchlor gel to affected skin areas, the mechlorethamine penetrates the tissue and interferes with cancer cell DNA replication. This mechanism proves particularly effective against mycosis fungoides, the most common type of cutaneous T-cell lymphoma, where malignant T-cells migrate to the skin and cause lesions to appear.
You’ll appreciate that this targeted approach addresses a critical medical need. Mycosis fungoides has no cure, and its cause remains unknown. The condition progresses from initial rash-like appearances to potentially disfiguring tumors. More concerning, if left untreated, these affected T-cells can migrate into the lymph system, spreading the cancer throughout your body.
The alkylation process you’re learning about represents a fundamental cancer-fighting mechanism that prevents these malignant cells from successfully dividing and spreading. Professor Crooks emphasized the importance of early intervention, noting that with proper diagnosis and treatment using mechlorethamine, you can potentially prevent the progression of cutaneous T-cell lymphoma.
The Birth of Over 100 Life-Saving Chemotherapy Drugs
Previously, I’ve outlined how one wartime discovery led to mechlorethamine’s development, but you should understand that this represents just the beginning of a pharmaceutical revolution. The principles discovered through nitrogen mustard research laid the foundation for an entire class of chemotherapy medications.
The alkylating mechanism you’ve learned about became the template for developing numerous other cancer treatments. Each new drug built upon the fundamental understanding that emerged from those initial wartime medical discoveries, creating a comprehensive arsenal against various forms of cancer.
When you consider Professor Crooks’ ongoing research, you’ll see how this legacy continues. He’s applying the knowledge gained from Valchlor’s development to create new treatments for acute myelogenous leukemia (AML), the most common form of leukemia, and even Alzheimer’s disease. This demonstrates how wartime pharmaceutical research continues influencing modern drug development across multiple therapeutic areas.
Your understanding of this progression reveals how one tragic wartime discovery became the cornerstone for countless life-saving medications, transforming cancer care from a field with limited options to one offering hope through targeted, scientifically-based treatments.
Revolutionary Advances in Modern Cancer Treatment
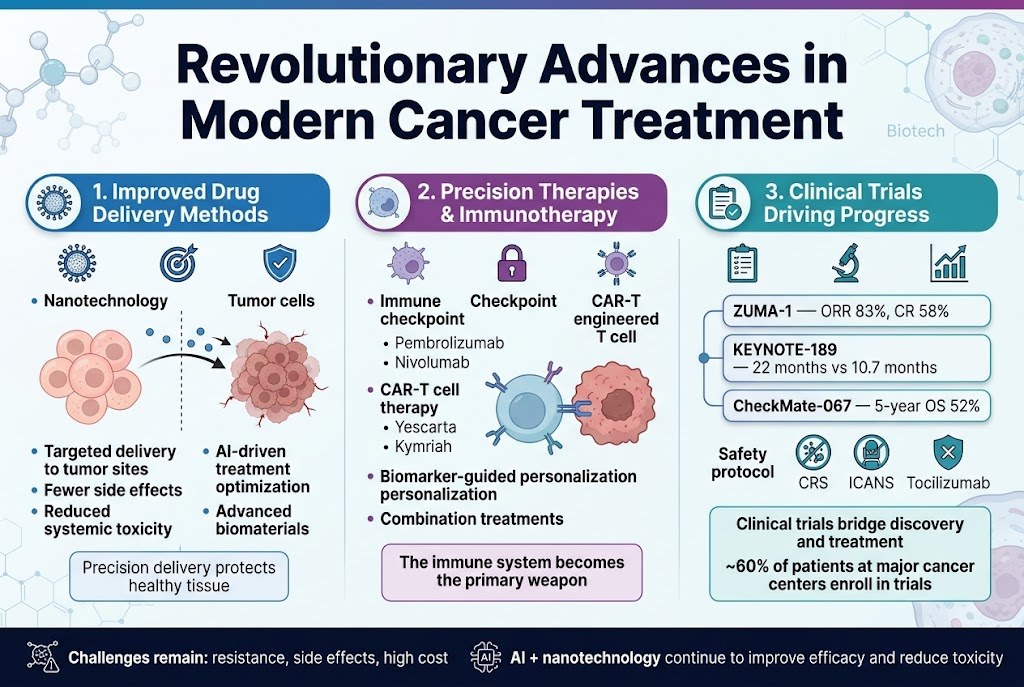
Improved Drug Delivery Methods and Reduced Side Effects
Now that we understand how wartime discoveries laid the foundation for modern cancer treatment, you’ll see how today’s revolutionary advances have transformed the therapeutic landscape through sophisticated delivery mechanisms. Modern cancer treatment has evolved to address one of chemotherapy’s most significant challenges: the indiscriminate targeting of both healthy and cancerous cells that plagued early treatments derived from wartime chemical weapons.
You’re witnessing a paradigm shift in how cancer drugs reach their targets. Nanotechnology has emerged as a game-changing approach, enabling precise drug delivery that minimizes damage to healthy tissues. These advanced delivery systems utilize engineered biomaterials and nanoparticles to transport therapeutic agents directly to tumor sites, dramatically reducing the systemic toxicity that you might have experienced with traditional chemotherapy.
The development of targeted delivery methods represents a stark contrast to the broad-spectrum destruction of early chemical weapon-derived treatments. You can now benefit from therapies that selectively target cancer cells while sparing surrounding healthy tissues, resulting in significantly fewer side effects and improved quality of life during treatment. These precision delivery systems have transformed the patient experience from the harsh realities of early chemotherapy to more tolerable and effective treatments.
Artificial intelligence and advanced biomaterials are revolutionizing how you receive cancer treatment. These technologies enable clinicians to design customized treatment regimens that optimize drug delivery while minimizing adverse effects. The integration of AI-driven treatment optimization helps predict how your body will respond to specific delivery methods, allowing for personalized approaches that maximize therapeutic benefit.
Precision Therapies and Immunotherapy Breakthroughs
With these delivery improvements in mind, you’re now entering an era where your immune system becomes the primary weapon against cancer. Immunotherapy has revolutionized cancer treatment by harnessing your body’s natural defenses to identify and destroy cancer cells, offering a more targeted and often more effective approach than conventional methods.
You can now access immune checkpoint inhibitors that work by blocking proteins cancer cells use to evade your immune system. These breakthrough treatments, including pembrolizumab and nivolumab, have shown remarkable success in treating cancers like melanoma and non-small cell lung cancer, where traditional therapies often failed. The success of pembrolizumab in advanced melanoma treatment demonstrates how these therapies can achieve long-term remission in patients who previously had limited options.
CAR-T cell therapy represents another revolutionary approach available to you, particularly if you’re battling blood cancers. This treatment involves genetically modifying your own T cells to express chimeric antigen receptors, dramatically enhancing their ability to detect and destroy cancer cells. You’ll find that CAR-T therapies like Yescarta and Kymriah have achieved unprecedented success rates, especially in cases resistant to other treatments.
Your treatment options now include personalized immunotherapy guided by biomarkers, genomic data, and tumor environment analysis. This precision approach enables clinicians to tailor treatments to your specific tumor characteristics, improving response rates while reducing unnecessary side effects. You benefit from combination treatments that integrate immunotherapy with chemotherapy, radiation, and targeted therapies, demonstrating synergistic benefits that improve your overall outcomes.
Despite these advances, you should understand that challenges such as resistance, side effects, and high treatment costs persist. However, ongoing technological innovations in nanotechnology and artificial intelligence continue to enhance treatment efficacy while reducing toxicity, making these life-saving therapies increasingly accessible to you.
The Critical Role of Clinical Trials in Drug Development
Previously, we’ve seen how precision therapies emerged from rigorous research, and you’ll now discover how clinical trials serve as the crucial bridge between laboratory discoveries and the treatments available to you today. Your access to these revolutionary cancer treatments depends entirely on the systematic evaluation provided by clinical trials, which have shaped every major immunotherapy breakthrough you can now access.
You should understand that landmark trials like ZUMA-1, JULIET, and CheckMate have directly impacted the treatment options available to you. The ZUMA-1 trial, which evaluated axicabtagene ciloleucel in patients with relapsed/refractory large B-cell lymphoma, achieved an overall response rate of 83% with a complete response rate of 58%, demonstrating the remarkable efficacy that you might experience with CAR-T therapy.
Clinical trials have been essential in establishing safety protocols that protect you during treatment. You benefit from standardized management protocols for conditions like cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS) that were developed through careful clinical observation. These trials identified that tocilizumab, an anti-IL-6 receptor antibody, effectively manages CRS, directly improving your safety during CAR-T therapy.
Your treatment options continue expanding through ongoing clinical trials that evaluate combination therapies. The KEYNOTE-189 trial demonstrated that combining pembrolizumab with chemotherapy resulted in a median overall survival of 22 months versus 10.7 months with chemotherapy alone, providing you with significantly better outcomes. Similarly, the CheckMate-067 trial showed that combining nivolumab and ipilimumab achieved a 5-year overall survival rate of 52% in advanced melanoma patients.
You should know that participation in clinical trials often provides access to cutting-edge treatments before they become widely available. Approximately 60% of patients visiting major cancer centers are enrolled in clinical trials, often with no drug costs to participants. This participation not only advances medical knowledge but also provides you with potential access to breakthrough treatments that might not otherwise be available.
The rigorous evaluation process of clinical trials ensures that when new treatments become available to you, they have been thoroughly tested for both efficacy and safety, representing the culmination of years of careful research that began with those early wartime discoveries.
Uncovering Lost Medical History and Setting Records Straight

The Search for Patient JD’s Missing Medical Records
With the understanding of how wartime medical discoveries transformed modern cancer care, you might wonder how we know so much about these early breakthrough treatments. The story of patient J.D., the first person to receive intravenous chemotherapy, nearly remained lost to history forever. Your ability to learn about this pivotal moment in medical history exists only because of an extraordinary detective effort that took place nearly 70 years after the original treatment.
When J.D. received his groundbreaking nitrogen mustard treatment at Yale in 1942, his medical records seemed destined for obscurity. In the chaos of those war years, his complete medical file was misplaced, leaving only scattered memories and incomplete accounts. By 1946, when researchers attempted to document this historic case in the Journal of the American Medical Association, they had to rely on personal recollections rather than actual medical documentation. This resulted in numerous inaccuracies about J.D.’s treatment timeline, his medical condition, and even basic details about his life.
You can appreciate the magnitude of this challenge when you consider what Dr. John Fenn and Dr. Robert Udelsman faced two years ago when they became fascinated by J.D.’s case. These Yale physicians were determined to locate his original medical records, but they had virtually nothing to work with. They possessed only his initials – “J.D.” – with no name, date of birth, medical record number, or precise treatment dates. Your understanding of their persistence becomes clear when you realize they spent months methodically sifting through records of every “J.D.” they could find from the early 1940s.
Their search process involved examining pathology reports and medical files for dozens of patients with matching initials. When they finally discovered one J.D. with lymphosarcoma, they encountered another obstacle – the medical record number contained errors. Thanks to a persistent archivist who painstakingly sequenced and corrected the numbering system, they narrowed their possibilities down to one patient. After this exhaustive investigation, J.D.’s complete medical records were finally recovered.
Revealing the Personal Story Behind Medical History
Now that you understand how these records were recovered, you can appreciate the deeply personal story that emerged from J.D.’s file. Your glimpse into his life reveals a man whose courage shaped medical history, yet whose personal circumstances were marked by profound loneliness. Born in Poland in 1894, J.D. immigrated to the United States at age 18, seeking the opportunities that America promised to so many.
You discover through his medical records that J.D. lived a solitary existence in a rented four-room house in Meriden, Connecticut. He never married, had no known family nearby, and worked steadily at a ball-bearing factory until his illness began in 1940. When lymphosarcoma – a malignant tumor of his lymph nodes – began as enlarged tonsils and progressed to massive tumors around his neck, J.D. faced his battle largely alone.
Your understanding of his isolation becomes even more poignant when you learn about his hospital experience. Throughout his 96-day stay at New Haven Hospital, J.D. received visitors only once, near the end of his life. The nurse’s notation in his file reveals the heartbreaking reality of his situation: those visitors asked to be notified only if his condition improved, but stated they would not return otherwise. Dr. Fenn, one of the physicians who recovered J.D.’s records, described this discovery as heartbreaking, noting how it revealed the profound loneliness of J.D.’s final months.
Despite facing terminal illness and social isolation, you can see J.D.’s remarkable courage when Dr. Gustaf Lindskog approached him about participating in the experimental nitrogen mustard treatment. Knowing he was dying and understanding the experimental nature of the treatment, J.D. willingly agreed to become the first human recipient of intravenous chemotherapy. Your appreciation for his decision grows when you realize he had no family support system and was essentially trusting his life to an unnamed chemical that doctors could only refer to as “substance X” due to wartime secrecy requirements.
Recognizing Pioneering Researchers Who Changed Cancer Treatment Forever
Previously, you’ve learned about J.D.’s personal story, but understanding the complete picture requires recognizing the brilliant researchers whose collaboration made this breakthrough possible. Your knowledge of cancer treatment history would be incomplete without acknowledging the team of Yale scientists who transformed a deadly chemical weapon into a life-saving medical treatment.
The pioneering work began with Drs. Louis Goodman and Alfred Gilman from Yale’s pharmacology department, working alongside Dr. Thomas Dougherty from anatomy. You should understand that their research was conducted under extraordinary circumstances – they were participating in a top-secret U.S. government program to develop antidotes to chemical warfare agents, fearing German use of such weapons against Allied soldiers.
Your appreciation for their scientific methodology becomes clear when you examine their careful progression from laboratory to clinical application. Working with nitrogen mustard, they first treated a mouse with advanced lymphosarcoma, observing remarkable initial results after just two injections. The tumor began to soften and reduce in size, surprising and exciting the research team. Although the cancer eventually developed resistance to treatment, the chemotherapy extended the mouse’s life for 84 days – a result they considered remarkable enough to justify human trials.
Dr. Gustaf Lindskog, an assistant professor of surgery at Yale, became the crucial bridge between laboratory research and clinical application. You can see his vital role when the research team approached him to find a suitable cancer patient for the first clinical trial of nitrogen mustard. His willingness to present this experimental treatment option to J.D. demonstrated the collaborative spirit that made this breakthrough possible.
With this understanding of the research team’s contributions, you can appreciate how their work established fundamental principles of chemotherapy that remain relevant today. Dr. Fenn noted that J.D.’s case contained “the revelation that cancer would respond to chemical injections, and that chemotherapy also had potentially lethal implications in the depression of bone marrow.” Dr. Robert Udelsman emphasized that “that first proof of chemotherapy’s efficacy as well as its side-effects paved the way for decades of improvement in these life-saving agents.”
Your recognition of these researchers’ contributions helps set the historical record straight about the birth of modern cancer treatment. Their careful documentation, despite wartime censorship requirements, and their willingness to take calculated risks with experimental treatments, established the foundation for medical oncology that has since saved millions of lives worldwide.
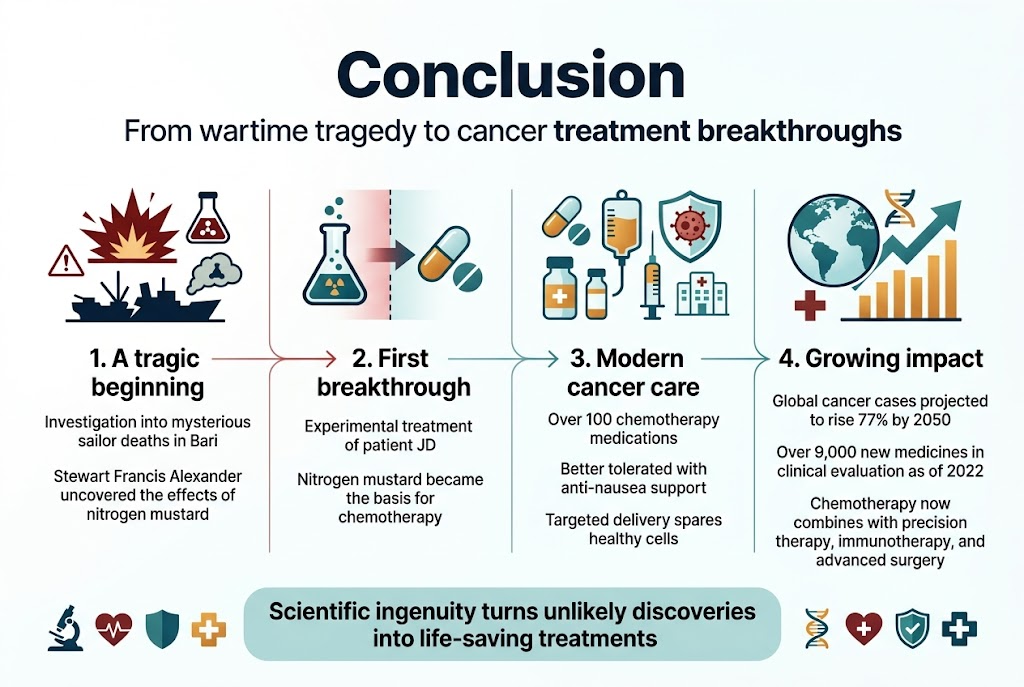
The journey from a tragic wartime disaster to life-saving cancer treatment reveals how scientific breakthroughs can emerge from the most unexpected circumstances. What began as Lieutenant Colonel Stewart Francis Alexander’s investigation into mysterious sailor deaths in Bari has evolved into a sophisticated arsenal of over 100 chemotherapy medications that continue to transform cancer care worldwide. Today’s treatments are far more tolerable than early nitrogen mustard trials, thanks to advanced anti-nausea medications and innovative delivery methods that target tumors while sparing healthy cells.
As cancer cases are projected to increase 77% worldwide by 2050, the legacy of these wartime discoveries becomes even more critical. The accelerating pace of drug development—with over 9,000 new medicines in clinical evaluation as of 2022—demonstrates how far you’ve come from that first experimental treatment of patient JD. Modern cancer centers now combine chemotherapy with precision therapies, immunotherapies, and advanced surgical techniques, offering you hope where once there was none. The transformation of a deadly chemical weapon into a cornerstone of cancer treatment stands as a testament to scientific ingenuity and reminds you that breakthrough discoveries often come from the most unlikely sources.

